WeightWatchers Weight-Loss Program—Lose Weight and Keep it Off | WW Canada
Ends today: 6 months free! With select plan purchase.
A totally “worth it” thing you can do for your health
WW gives you a science-backed, eat-real-food, flexible weight-loss program that works.


6 months free
Your future self will thank you!


For weight loss to work, it has to work for you
Prioritizing your health is challenging when work is stressful, schedules are packed, and pizza delivery is a tap away.
We help you overcome obstacles that get in the way of weight loss.
What WeightWatchers® is all about
/ Nutrition made simple /
Life is complicated—healthy eating shouldn’t be. Our weight-loss program and customized plans make it easier.
/ No quick fixes /
We’re not a deprivation diet or prepackaged meals. We’re nutritional and behavioral science that changes your relationship with food—so you can keep your favorites.
/ A strong support system /
You don't have to do it alone. Our expert coaches and millions of members understand weight loss and are here to support you—online or in person.
/ Nutrition made simple /
Life is complicated—healthy eating shouldn’t be. Our weight-loss program and customized plans make it easier.
/ No quick fixes /
We’re not a deprivation diet or prepackaged meals. We’re nutritional and behavioral science that changes your relationship with food—so you can keep your favorites.
/ A strong support system /
You don't have to do it alone. Our expert coaches and millions of members understand weight loss and are here to support you—online or in person.
How it works
(Hint: You’ll eat what you love)
We use a program no one else does, so you’ll lose weight—not your favourite foods.


/01
Meet the Point
The Points® system takes a food’s specific nutritional info and turns it into a single number.
/02
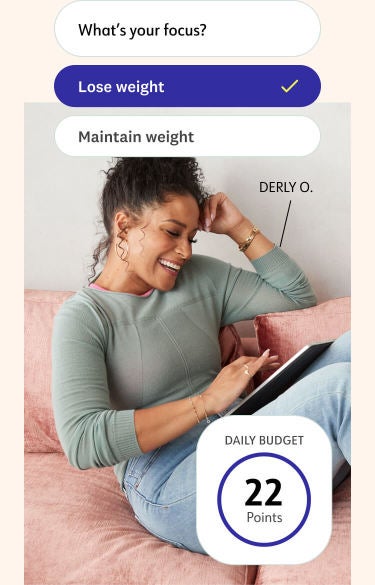
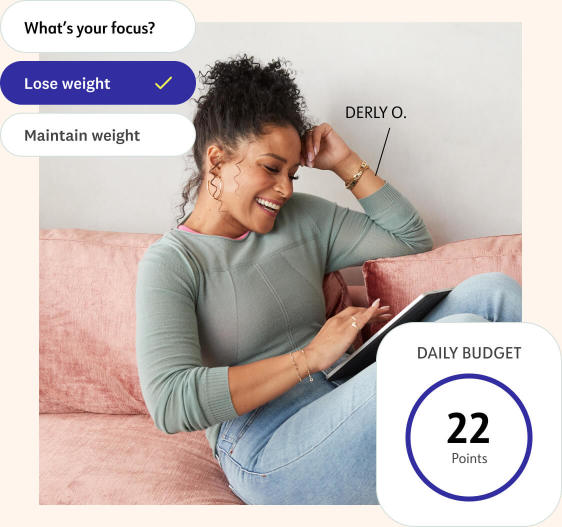
Get your plan
After you join, answer some quick questions in our app. You'll immediately get a nutrition plan and Points to "spend" each day.

/03
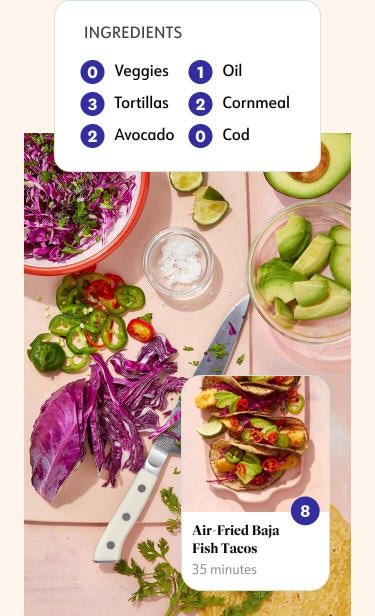
Make healthier choices
Points guide you to eat more nutritious foods and help you find portion sizes that are right for you.

/04
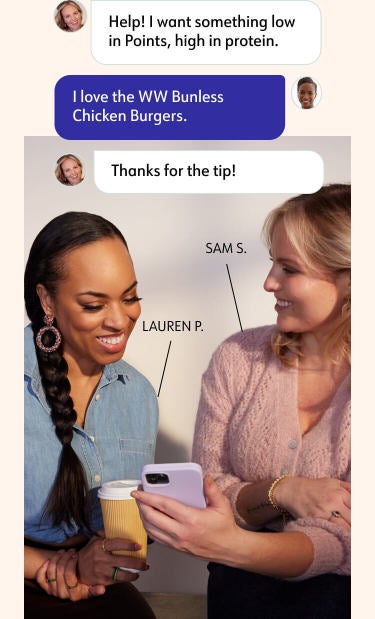
Find your community
Meet members face-to-face or virtually at Workshops, and tap into our exclusive social network, 24/7. Share tips, recipes, and solutions for common challenges.
Everything you need for an easier journey

Results you can see. Health you can feel.
Take it from our members.
Stay in the loop
Frequently asked questions
WeightWatchers is the #1 doctor-recommended weight-loss program†. Additionally, our diabetes-tailored plan is based on guidelines from the American Diabetes Association and the International Diabetes Federation.
† Based on a 2020 IQVIA survey of 14,000 doctors who recommend weight-loss programs to patients.
Generally, memberships start for as low as $23 per month, depending on length of commitment you choose. See offers and pricing here.
We help you lose weight and build healthy habits, one small step at a time. Fad weight-loss diets can be restrictive, and rarely work long term. We have more than 140 publications that show WeightWatchers’ efficacy to help members lose weight and keep it off, and we’ve been ranked the #1 Best Diet for Weight Loss by U.S. News & World Report for 13 years in a row.†† See how WeightWatchers stacks up against the competition.
†† U.S. News & World Report ranked WW the #1 Best Diet for Weight Loss for 13 years in a row (2011–2023).
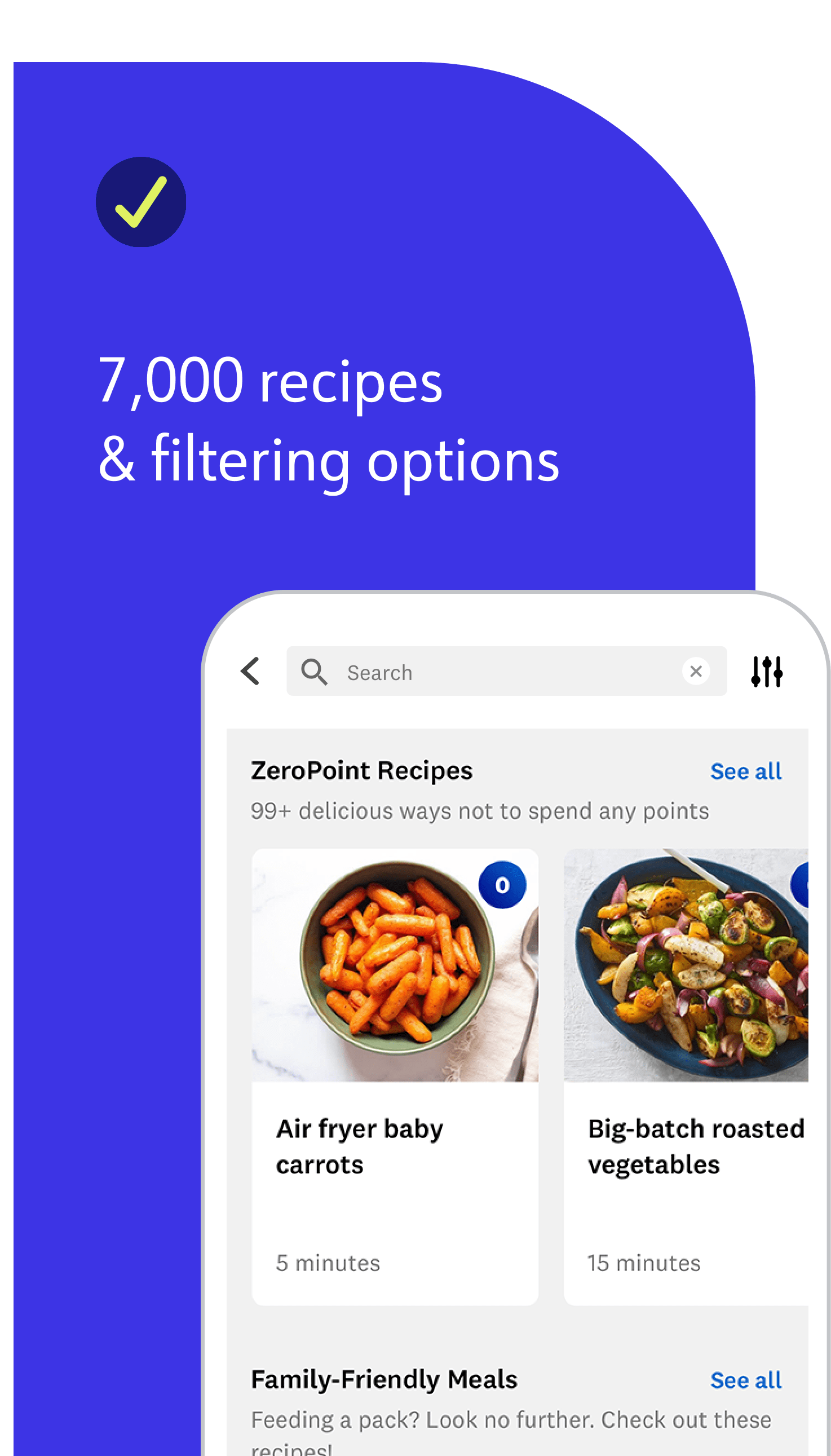
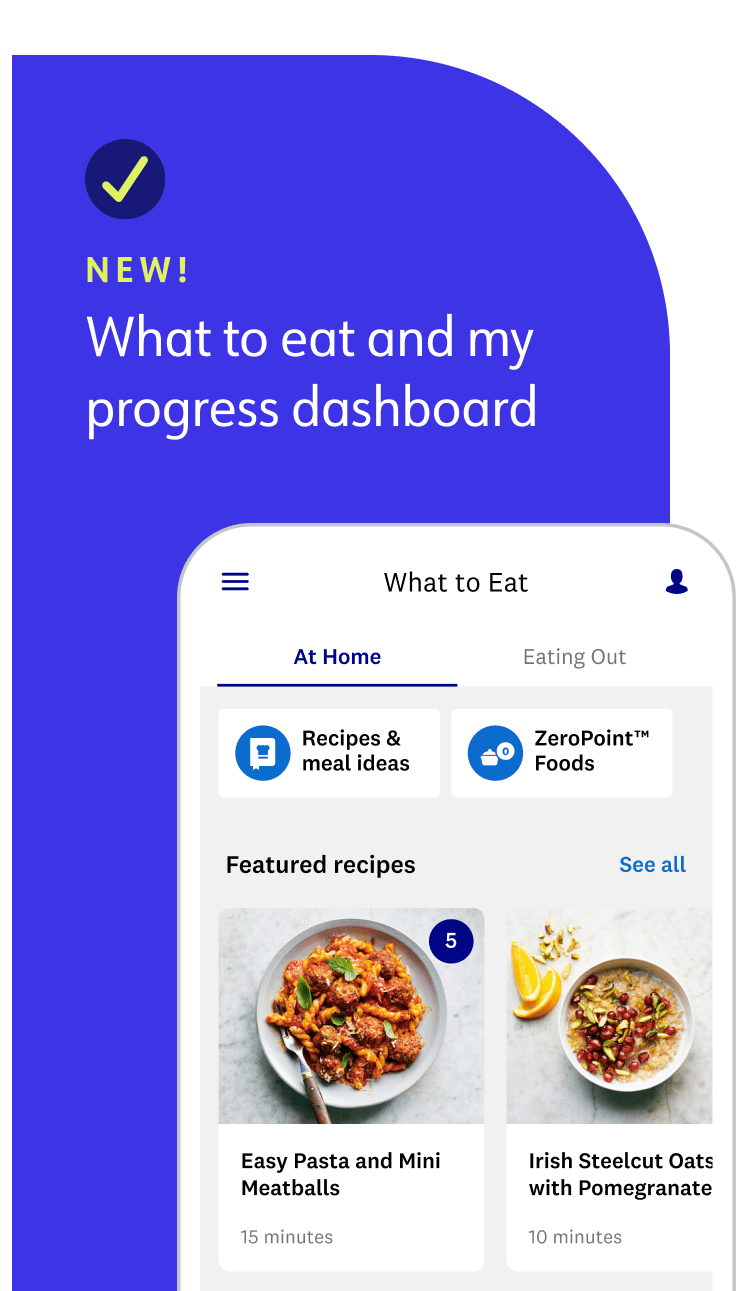
Our updated, simplified weight-loss program makes it easier for you to follow and find success. Every member gets a tailored Points Budget and streamlined list of over 200 ZeroPoint foods. Compared to programs of the past, we now use advanced nutrition science to factor in more food label data, like added sugars, fiber, and unsaturated fats, to guide you toward the healthiest foods.
What you eat is up to you—and we’ll show you how to enjoy your favourite foods in a way that gets you to your goals. With WeightWatchers, there are no “good” or “bad” foods—just nutritious and less nutritious ones.
We make it as easy as possible. You’ll get a Points® Budget to “spend” however you want. You’ll track your meals, drinks, and snacks in the WW app and enjoy as many ZeroPoint™ foods as you'd like. Activity can also boost your weight loss (vs. only focusing on food)ᶿᶿ.
ᶿᶿ Johns DJ, Hartmann-Boyce J, Jebb SA, Aveyard P; Behavioural Weight Management Review Group. Diet or exercise interventions vs combined behavioral weight management programs: a systematic review and meta-analysis of direct comparisons. J Acad Nutr Diet. 2014;114(10):1557–1568. doi:10.1016/j.jand.2014.07.005
ᶿDF Tate et al, 12-month randomized multi-country trial comparing weight loss between the WW program to a Do-It-Yourself approach where resource guides were provided. JAMA Network Open. 2022;5(8):e2226561. Funded by WW International, Inc.
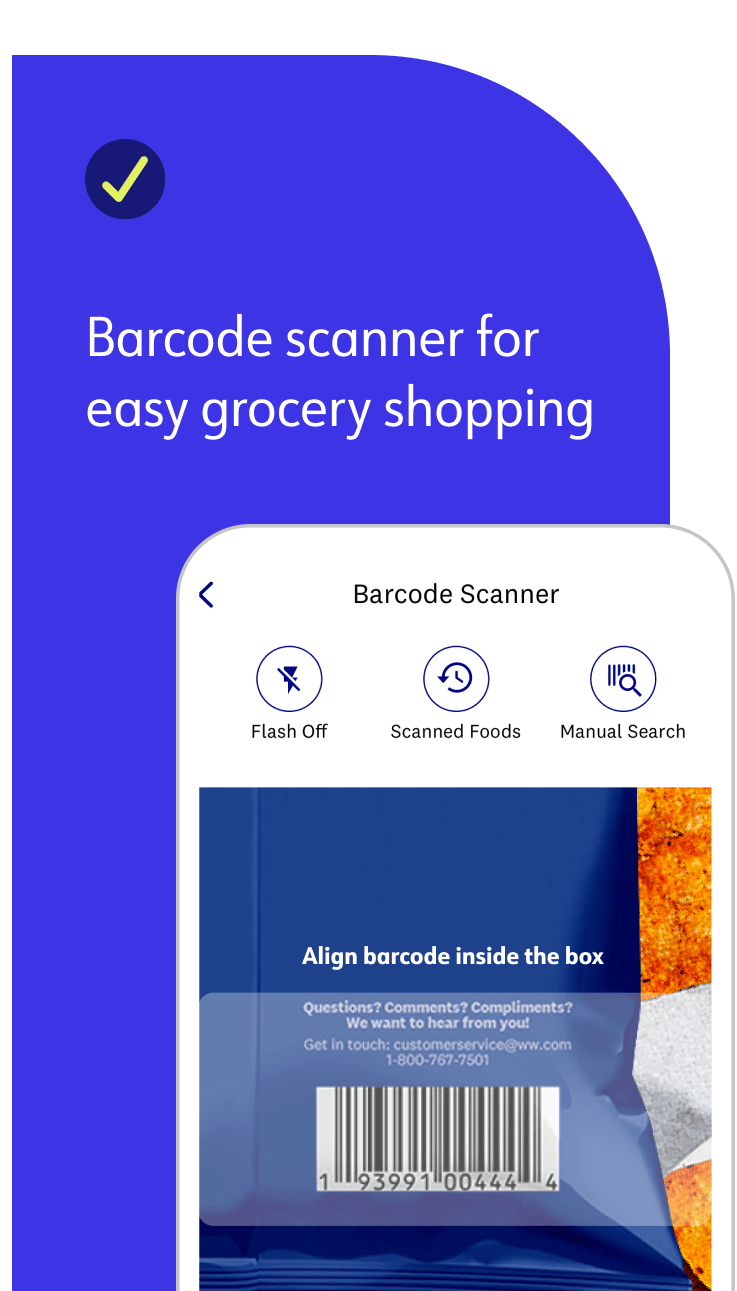
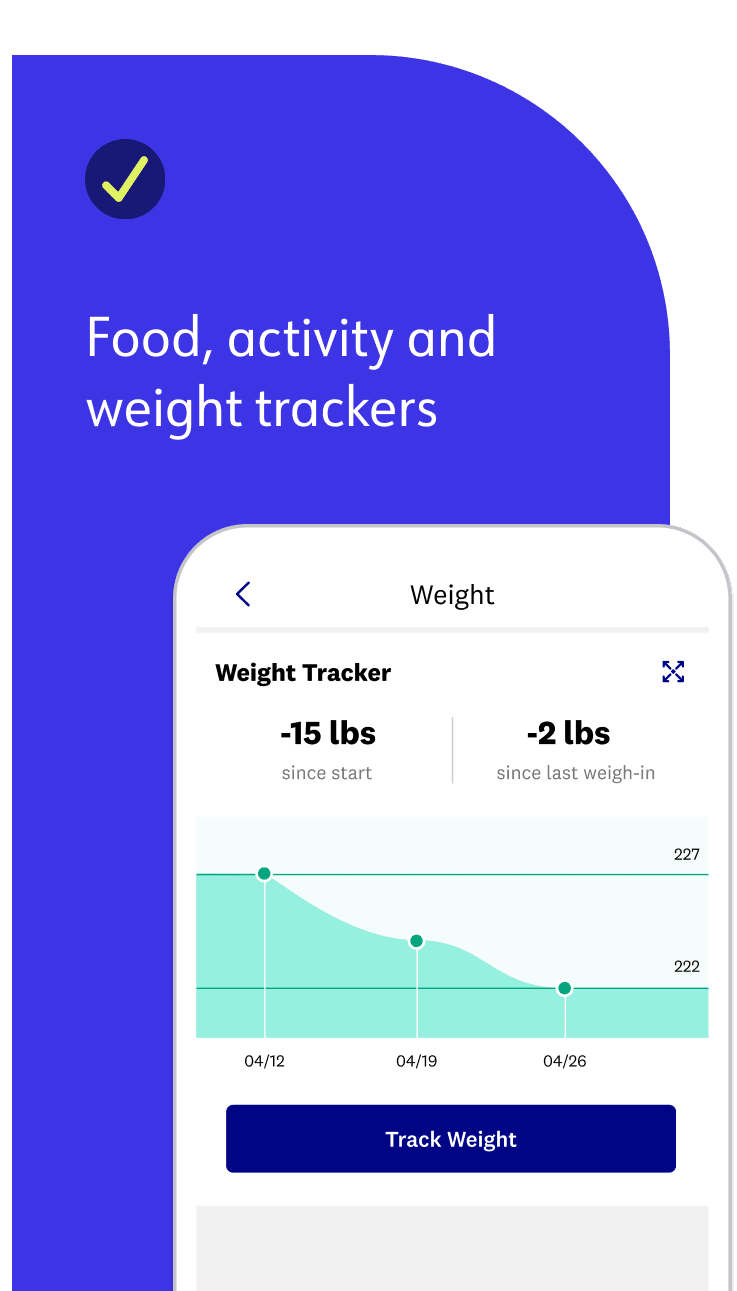
Science shows food tracking is the biggest predictor of success—the more you track, the more weight you’ll lose! But we’re all about consistency over perfection: It’s better to track some food most days of the week than every single thing you eat only some days of the week. In other words, perfection is not required. You’ll quickly decide how to approach tracking in a way that works for you. Here are some of our best tracking tips.
They are nutritional powerhouses that serve as the foundation of healthy eating and support your overall health. ZeroPoint foods also add bulk and flavour to meals, without costing Points® from your Budget, so you don’t have to weigh, measure, or track these foods—no matter how many you eat in a day! Those 200-plus foods include non-starchy veggies, fruit, eggs, yogurt, cottage cheese, fish, shellfish, chicken and turkey breast, and more.
Our team includes nutrition and behavioral scientists, registered dieticians, clinical researchers, and more. Together they help you overcome common weight-loss hurdles—like stress-eating or late-night snacking—while our exclusive Points system makes nutrition easier—so easy that healthier eating will feel like second nature. After you sign up, you’ll answer a few questions to get a plan customized for you, and build healthy habits with proven behavior change techniques.
Our Workshops (or Meetings, back in the day) are coach-led group sessions held in a Studio location or online. You don’t need to go, but the extra support can ramp up your results: Workshop members lose two times more weight than those who DIY.^ Workshops offer unlimited accountability, and give you a sense of belonging alongside coaches and members who really get it.
^^Based on a 6-month multicenter trial. Apolzan JW, et al. A Scalable, Virtual Weight Management Program Tailored for Adults (n=136) with Type 2 Diabetes: Effects on Glycemic Control. Presented at American Diabetes Association’s 82nd Scientific Sessions. 2022.
We know living with diabetes is challenging. Our diabetes-tailored plan for weight loss helps you lower blood sugar^^, lose weight, and stress less**.
^^Based on a 6-month multicenter trial. Apolzan JW, et al. A Scalable, Virtual Weight Management Program Tailored for Adults (n=136) with Type 2 Diabetes: Effects on Glycemic Control. Presented at American Diabetes Association’s 82nd Scientific Sessions. 2022.
**Based on a 6-month multicenter study demonstrating significant reductions in diabetes-related distress. Apolzan JW, et al. A Scalable, Virtual Weight Management Program Tailored for Adults with Type 2 Diabetes: Effects on Glycemic Control. Presented at American Diabetes Association’s 82nd Scientific Sessions. 2022.